Innovation drives everything
Promoting progress in clinical practice. Carrying out clinical research.
Realizing research cooperation with external partners. Investing in multi-center studies. Bundling new insights and knowledge. Being a forum for experts and the voice of our field of expertise. Innovation really drives things forward at Dr. Falk Pharma. Today as it always has.
Research and Development
The core here is the many years of experience in the areas of pharmaceutical, preclinical and clinical development. The teams of Dr. Falk Pharma implement the development projects in close cooperation with contract research organisations.
In galenics, Dr. Falk Pharma has developed outstanding expertise in topically - i.e. locally - effective drugs for the intestinal tract and the esophagus.
This has led and will lead to a whole host of patents and clinical approvals.
Multi-center studies play an essential role in the approval of new drugs.
The teams of Dr. Falk Pharma are very experienced in organizing large multi-center studies and have been able to establish an international network of clinical investigators over many years. This means that even extensive studies can be carried out quickly and successfully.
Investment in three dimensions
The aim is to further expand the range of therapeutic options in the field of digestive and metabolic medicine.
The further development of existing forms of therapy:
Areas in which the company is a leader and is successful.
For example with new galenics for existing products.
The launch of new forms of therapy:
Currently, Dr. Falk Pharma is the first to offer a product for the clinical condition of Eosinophilic Esophagitis (EoE): a chronic inflammatory, immune-mediated disease of the esophagus that leads to swallowing difficulties.
The development of new indication areas:
Dr. Falk Pharma is therefore investing significantly in studies researching into new active agents for clinical indications which need far more effective therapies: such as primary sclerosing cholangitis (PSC), metabolic dysfunction associated steatohepatitis (MASH)* and celiac disease or gastroparesis.
Partnering – stronger together
We continuously strive for transforming ideas and current advances in medical treatment. Dr. Falk Pharma cooperates with reliable partners in order to develop and implement high potential innovative treatment concepts. These can arise from any pharmacological concept: from “small molecules” to biologics. Within these projects, a wide variety of developmental steps are considered, which can have their origin in academic research, in clinical use, in start-ups or even established companies.
We look forward to making new contacts and establishing new partners, both nationally and internationally, in the fields of digestive and metabolic medicine. Feel free to contact us directly.
Current research and development activities
The focus of our current development projects covers both new drugs for inflammatory bowel disease, other bowel disorders, hepatic, biliary, and esophageal disorders, as well as the continuing development of previously-approved drugs - for example through the use of improved formulations. We are also investigating new therapeutic indications for currently-approved drugs.
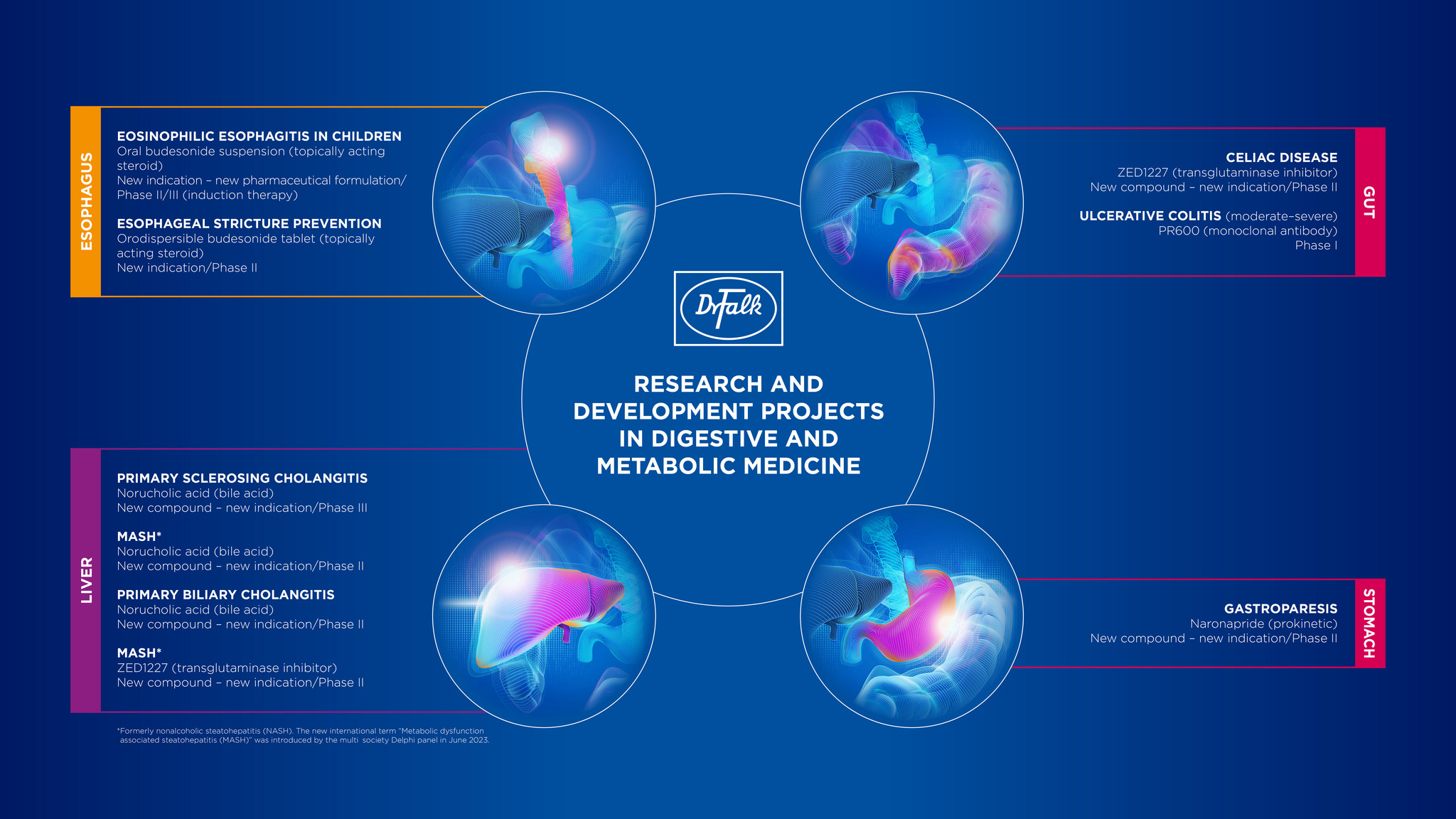
The following research projects are the particular focus of our present work:
- Ulcerative colitis: PR600 (mAb)
For more detailed information on the conditions of ulcerative colitis, please visit the indications page.
- Celiac disease: ZED1227
- Gastroparesis: Naronapride
- Primary sclerosing cholangitis: Norucholic acid
- Primary biliary cholangitis: Norucholic acid
For more information on primary biliary cholangitis, click here.
- Metabolic dysfunction associated steatohepatitis (MASH)*: Norucholic acid & ZED1227
' Formerly nonalcoholic steatohepatitis (NASH). The new international term "MASH" was introduced by the multi‐society Delphi panel in June 2023.
- Pediatric eosinophilic esophagitis (under age 18): Budesonide suspension
- Esophageal stricture prevention: Orodispersible budesonide tablets with effervescent properties
For more details on eosinophilic esophagitis, click here.
An overview of our entire research and development program can be found at EudraCT or in the graphic below.
* Formerly nonalcoholic steatohepatitis (NASH). The new international term "MASH" was introduced by the multi‐society Delphi panel in June 2023